
Innoo Tech Misting Cooling System 75.4FT (23M) Misting Line + 34 Upgrade Brass Mist Nozzles + a Brass Adapter(3/4") Outdoor Mister for Patio Garden Greenhouse Trampoline for waterpark
- 💧 Upgraded high quality: Precision brass spray nozzle, precision production, made of high quality UV treatment hose, UV resistance up to 3.5 or higher, yellowing resistance, hydrolysis resistance, microbial degradation resistance.
- 💦 Easy to install: With fixed straps and buckles, can be easily attached to trampolines and parasols. The fully assembled system can be installed in less than 10 minutes, providing very fine mist in hot summer weather. To significantly cool the air.
- 🌊 Cooling: The outdoor cooling system provides a cool environment for your favorite outdoor area, reducing the ambient air temperature to 20 degrees.
- 🏊 Energy saving and environmental protection: The atomizing cooling system is directly cooled by tap water spray, does not consume electricity, does not consume energy, saves you money and becomes an environmentalist.
- 🏄 Wide application: atomization cooling system is widely used in gardens, trampoline water parks, landscaping, greenhouses, humidification, outdoor cooling systems, epidemic prevention, spray disinfection, deodorizing air purification, swimming pool atomization cooling, atomizing mosquito systems, etc.
Retail Price:
$
35.99
Wholesale Price:
Inquire Now
This website follows the safe harbor principle. If any infringement, please inform us in time
and we will delete it in time.
About safe harbor,please click here to learn more.
After Sale Service:
1 Insurance Policy:
Click to go to the Help Center to view the Insurance Policy
2 The rules apply and summit for receive compensation
3 The conditions of accrual for amount insurance
3 The conditions of accrual for amount insurance
5 Damage to the parcel during international shipping, which resulted in damage to the goods in it
6 Loss of individual goods under international shipment
The absence individual goods in the parcel
7 Non-compliance individual goods in color, size, complications
Damage to the goods during international shipment
8 Returns Policy:
Click to go to the Help Center to view the Returns Policy
Innoo Tech Misting Cooling System 75.4FT (23M) Misting Line + 34 Upgrade Brass Mist Nozzles + a Brass Adapter(3/4") Outdoor Mister for Patio Garden Greenhouse Trampoline for waterpark
Innoo Tech Misting Cooling System 75.4FT (23M) Misting Line + 34 Upgrade Brass Mist Nozzles + a Brass Adapter(3/4") Outdoor Mister for Patio Garden Greenhouse Trampoline for waterpark
BEST SELLERS

Product Description
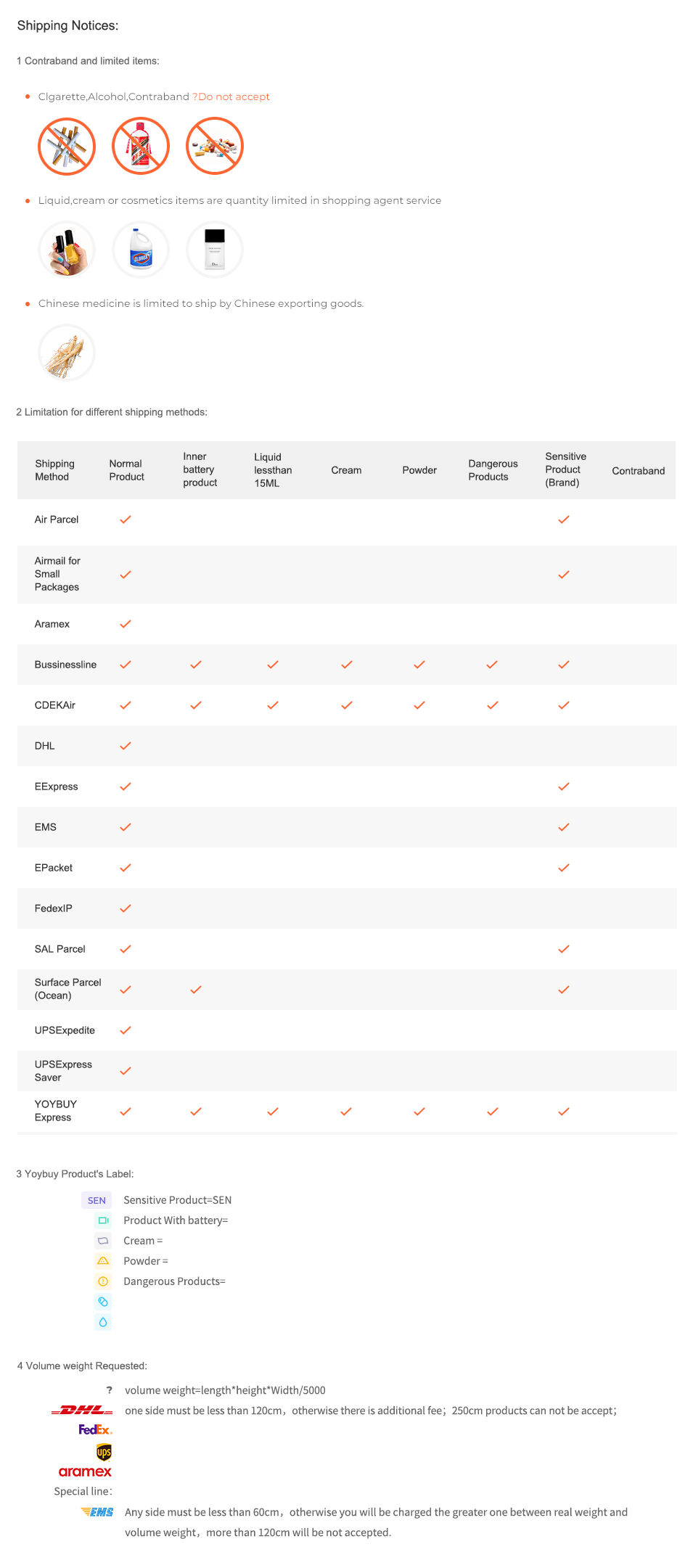
Shipping Notices:
1 Contraband and limited items:
Clgarette,Alcohol,Contraband ?Do not accept



Liquid,cream or cosmetics items are quantity limited in shopping agent service



Chinese medicine is limited to ship by Chinese exporting goods.

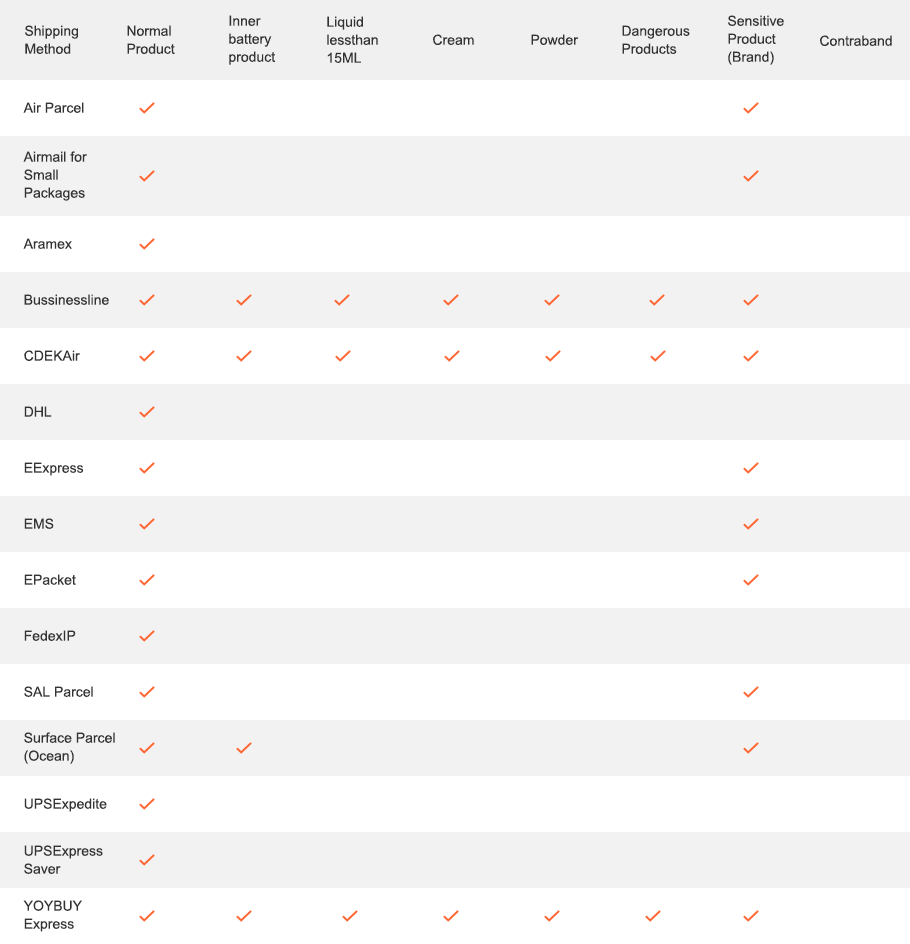
2 Limitation for different shipping methods:
3 Yoybuy Product's Label:
Sensitive Product(SEN)
Product With battery
Cream
Powder
Dangerous Products
4 Volume weight Requested:

volume weight=length*height*Width/5000

one side must be less than 120cm,otherwise there is additional fee;250cm products can not be accept;




Any side must be less than 60cm,otherwise you will be charged the greater one between real weight and volume weight,more than 120cm will be not accepted.
After Sale Service:
1 Insurance Policy: Click to go to the Help Center to view the Insurance Policy
2 The rules apply and summit for receive compensation
3 The conditions of accrual for amount insurance
3 The conditions of accrual for amount insurance
5 Damage to the parcel during international shipping, which resulted in damage to the goods in it
6 Loss of individual goods under international shipment
The absence individual goods in the parcel
7 Non-compliance individual goods in color, size, complications
Damage to the goods during international shipment
8 Returns Policy: Click to go to the Help Center to view the Returns Policy
BEST SELLERS


 Language
Language

 Currency
Currency
Upload image to find the same item
 Drag the image here, or
Click to upload
Tips: Only jpg, jpeg, png, webp
Drag the image here, or
Click to upload
Tips: Only jpg, jpeg, png, webp


Scan the QR code and use your phone to take a direct photo search.
×




















